Each year, hundreds of thousands of babies born prematurely in low- and middle-income countries die because medical facilities there cannot afford the equipment that could help babies survive those crucial first few weeks after birth.
Many of these deaths are caused by respiratory distress syndrome.
In sub-Saharan Africa alone, some 6 million preterm babies are born every year with immature lungs. Their lungs aren’t fully developed, and they have trouble staying inflated, so they collapse. While medical institutions in high-income countries have bubble continuous positive airway pressure machines to help them breathe, those bCPAP units cost thousands of dollars—making them prohibitively expensive for many low-income nations. Of those 6 million babies, 800,000 of them are born at mid-level facilities that require bCPAP devices but likely don’t have them.
The bCPAP devices keep the lungs from deflating and also deliver blended oxygen into them—a critical step because breathing 100% oxygen can cause blindness in premature babies.
Medical providers in some low-resource countries use improvised bCPAP kits assembled from parts they already have in their clinics and use them to help preterm babies survive. However, these kits do not have the ability to provide blended oxygen for babies.
A partnership to find a low-cost solution

To address this issue, a group of innovative medical providers and researchers from Seattle Children’s, the University of Washington, global health nonprofit PATH, Adara Development and Kiwoko Hospital in Uganda have partnered to create a low-cost device that could help thousands of premature babies breathe and survive.
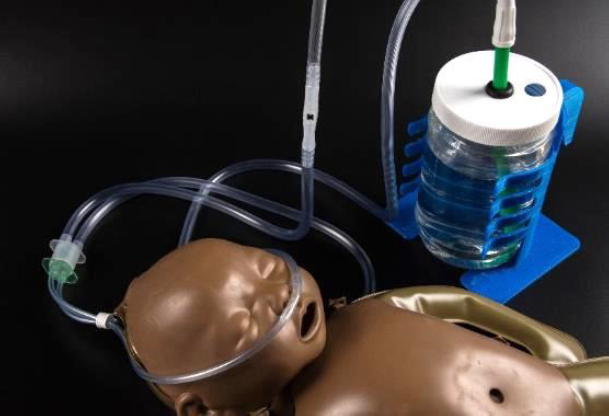
The low-cost bCPAP device has been tested at Kiwoko Hospital with funding from Saving Lives at Birth. It uses a series of tubes and a molded plastic piece that pulls in room air to blend the oxygen. It was designed by engineers at PATH in collaboration with the other partners.
Dr. Anna Hedstrom, Seattle Children’s and UW Medicine neonatologist and assistant professor in pediatrics at the University of Washington School of Medicine, focuses on global neonatal health and is part of the group that’s developing the low-cost bCPAP device. She works with sick or premature babies in intensive care environments where financial resources are scarce.
Hedstrom said that in hospitals in high-income countries, a provider can simply plug a line into a wall and get blended oxygen. However, in low-resource countries, medical staff only have access to 100% oxygen.
“The novel part of the low-cost bCPAP device is a component called the oxygen blender,” Hedstrom said. “This blender lets you use your source of oxygen and then deliver less than 100% oxygen to the baby, which reduces the risk for blindness.”
Hedstrom said that the science behind it is straightforward and equivalent to other devices that are already in use.
The low-cost bCPAP device combines room air with oxygen and delivers it to the baby’s nose. The tubing carrying the oxygen ends submerged in water, which creates the pressures in the system and makes bubbles when the air comes out. The bubbles create a vibration that helps to keep the lungs open and working better.
Hedstrom said the main issues the study tested in Uganda were the usability and feasibility of the device.
“The real question was, when you take a device like this into a very low-resource country in a setting where things are limited as far as availability, including nursing time — how does this device work when put in place?” said Hedstrom. “Nurses, who are the ones who provide the hands-on care for babies —their feedback was very positive, and they felt the bCPAP [devices] were very easy to use.”
The low-cost bCPAP device doesn’t need electricity, making it a good alternative for places where blackouts can happen frequently.
“It’s driven by flow, so that’s helpful for a setting in which electricity is not reliable,” Hedstrom said. “In the whole context to care for a premature baby, you need other things that do require electricity. It’s not entirely reasonable to think that this is something that can be used someplace with no electricity because you need other things for the baby. But it is helpful to have one less component that doesn’t require electricity.”
Global leadership
Hedstrom said Seattle Children’s interim neonatology chief Dr. Maneesh Batra’s leadership and research in global health was pivotal in establishing the partnership with Kiwoko Hospital that led to the study of the bCPAP device there.
Hedstrom’s interest in global neonatology health was based on her experience in medical school and residency. She said she’s working on projects in Kenya, Ghana and Ethiopia, but the COVID-19 pandemic has paused some of the in-country work.
“It’s actually helped a lot of our techniques for improving health and working with our collaborators,” Hedstrom said. “We haven’t actually traveled to these places, but we are able to facilitate better longitudinal work with them and help in a way that doesn’t involve flying in mission-style work, where you fly in for a couple weeks and fly out.”
Working on projects like the low-cost bCPAP device, which could save thousands of lives, is a big motivator for Hedstrom.
“This is what drives me. This is what gets me up in the morning and makes work exciting — to be able to translate this care to babies worldwide, instead of just having this very high level of care here in Seattle, which I also appreciate and is really important. It’s really what got a lot of us into this field,” she said. “It is taking all the knowledge that we have and then collaborating with people who are providing care to a whole different set of the population. We can learn from each other to move everything forward.”
Hedstrom added that she’s fortunate that she can focus on this work at Seattle Children’s along with a large group of global neonatologists and surrounding global health community who can support the work and make it successful.
An eye toward the future for the bCPAP device
Hedstrom said the group will keep working, and the feedback the team received from the study in Uganda will allow PATH engineers to refine the design.
“We know what worked and what didn’t,” she said. “What tube needs to be different length or different width or labeled a different way? Some of the device designs you can’t know until you actually try it.”
Ideally, Hedstrom said, the group developing the bCPAP device would receive grant funding to do more device development and conduct larger-scale studies in different environments. This would also support the manufacturing of the device and commercialize it, so providers can order it and use it.
She added that the group aims to have the product manufactured locally in Africa to ensure an affordable price and improve access in the facilities where it’s needed. There are manufacturers there who already make low-cost medical products for large markets.

