As the youngest in her family, Isabella Wallis is always following in the footsteps of her older siblings. While it has its perks in helping her learn the ropes, getting to be the first in her family at anything is few and far between.
That was until the 9-year-old from Olympia, Washington, became the first patient to enroll in a first-of-its-kind clinical trial at Seattle Children’s.
“It’s exciting to be able to help other people and give them more information about why they are sick,” said Isabella, who enrolled in the Precision Diagnostics in Inflammatory Bowel Disease, Cellular Therapy and Transplantation (PREDICT) trial after experiencing persistent inflammatory bowel disease (IBD) symptoms for more than six months.
When she didn’t feel well, Isabella, who had always been healthy and loved playing outside, would retreat to her bedroom, finding some comfort in watching reruns of her favorite show, The Simpsons, while lying down. But her upset stomach never went away.
Unclear what was causing Isabella’s illness or what treatment options may exist, her parents turned to Seattle Children’s at the advice of their pediatrician.
“The initial test results ordered by our pediatrician were all over the place,” said Jason Wallis, Isabella’s dad. “Some readings, such as her white blood cell count, were above normal. He immediately referred us to Seattle Children’s wanting to rule out any possible autoimmune disease.”
Improved diagnosis for more targeted treatment
The PREDICT trial Isabella is participating in aims to identify how the disease-fighting T cells of the immune system are disrupted in two groups of patients. One group includes those like Isabella who may have IBD. The second group involves patients who undergo bone marrow transplantation and who develop graft-versus-host disease (GVHD) of the gastrointestinal tract.
Both disorders are related to the immune system and both IBD patients and a subset of patients who have GVHD experience similar gastrointestinal symptoms, including vomiting, diarrhea and the inability to absorb nutrients.
Researchers will accomplish this by sequencing and looking for gene expression signatures – patterns of genetic alterations unique to the patient’s medical condition – in the T cells of 100 patients with IBD and 250 patients who have had bone marrow transplants. Ultimately, the research team hopes to uncover information that can enable molecular diagnoses of these disorders, as well as identify targeted therapies to better treat them.

“With the limited information available about why someone develops IBD or GVHD, patients are treated with a one-size-fits-all approach,” said Dr. Leslie Kean, associate director of the Ben Towne Center for Childhood Cancer Research and principal investigator of the trial. “There are a number of targeted therapies available, but we need more information to determine who should receive which therapy.”
A way to ‘put meaning behind her illness’
In May, Isabella came to Seattle Children’s for an endoscopy procedure so doctors could get a definitive look at what was happening in her intestines. At the same visit, blood and tissue samples were collected and sent to a lab for sequencing as part of the research study.
“Despite having many of the classic IBD symptoms, there was still a possibility that Isabella’s disease was not immune related,” said Dr. Ghassan Wahbeh, the gastroenterologist overseeing Isabella’s care and director of the Inflammatory Bowel Disease Center at Seattle Children’s. “This is often the case in children and completely changes the type of treatment we offer.”
Isabella’s mom, Shawna Wallis, was nervous about the endoscopy because it was the first time Isabella would need anesthesia. Knowing that they were contributing to the clinical trial helped her maintain perspective.
“Not knowing what is causing her to be sick is the worst,” said Shawna. “We want to be able to put meaning behind her illness and help other kids who might go through the same thing in the future.”
When the culprit is not IBD
During a recent follow-up visit with the Wallis family at Seattle Children’s South Clinic, Wahbeh explained that the endoscopy showed that Isabella’s symptoms were likely not caused by a change in her immune system. Rather, he describes her illness as a functional gastrointestinal disorder.
Functional gastrointestinal disorders, or FGIDs, are common in children and adolescents. Symptoms include chronic or recurrent gastrointestinal distress.
Unlike other bowel diseases, FGIDs do not result from structural or biochemical abnormalities. Instead, they are the result of dysfunction often linked to psychological or diet stressors.
They can significantly impact a child’s quality of life, causing them to miss out on school and other activities due to their symptoms. The PREDICT trial is also sequencing the cells of patients with FGID, with the hope of identifying the molecular causes of this disease as well.
“Patients with FGID will complain of very real GI distress, yet the endoscopy results will come back normal,” said Wahbeh. “This is what we saw with Isabella. Even with abdominal pain and diarrhea, her endoscopy showed no signs of inflammation.”
Sequencing millions of Isabella’s cells
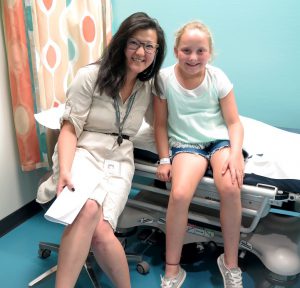
Dr. Betty Zheng, a gastroenterologist collaborating with Kean and Wahbeh on the PREDICT trial was also on hand to take another blood sample for analysis, and review the initial sequencing results with Isabella and her family.
Over 100,000 cells from Isabella’s endoscopy samples and 7 million cells from her blood work were analyzed as part of the PREDICT trial.
While PREDICT is a diagnostic clinical trial, meaning that its goal is to improve the pre-treatment information available to doctors and does not make treatment recommendations, researchers will be able to compare the genetic makeup of patients’ cells and begin to categorize them into various sub-types of bowel disease.
Zheng explained how the cells collected from Isabella will be part of the study’s control group and compared to those diagnosed with IBD.
“Working from the bedside backward to the bench is key to our study,” said Zheng. “Our hope with PREDICT is that we can better understand why some kids with pain experience inflammation and develop IBD and others do not. This clinical trial will hopefully help us provide critical information to patients and families through molecular blood tests and allow us to better formulate a treatment plan matched to their specific type of bowel disorder.”
Zheng noted how the team will apply the same approach to bone marrow transplant patients who develop GVHD of the gastrointestinal tract, making a difference in the diagnosis and treatment of that disease as well.
‘Why wouldn’t you help?’

Treatment for FGID focuses on making incremental changes to a child’s diet and teaching them to use cognitive behavioral therapy techniques so that they develop mind-body awareness and can self manage the stressors that lead to pain.
“We are taking small steps to help Isabella be mindful of what triggers her symptoms,” said Wahbeh. “Other little changes, such as how much she eats at one time and learning to eat more slowly by chewing her food completely, can make a difference too.”
Isabella and her dad, who does most of the family’s cooking, have started swapping soda for water and are looking for ways to add more vegetables to their meals. Isabella is already learning tricks for making her healthy snacks more appetizing. At the end of their visit, she asked Zheng, “Do you think it would be okay if I add peanut butter to my celery to make it a little sweet?”
Jason knows helping Isabella overcome her illness will be a process that the whole family will need to contribute to rather than an overnight fix. As a cancer survivor, he has been through his share of medical tests and treatments and sees the value of having the option to enroll Isabella in the trial.
“Dr. Wahbeh empowers Isabella to talk about her symptoms and give him information so that he can help her feel better,” said Jason. “Being a part of the research is also empowering in a way for Isabella. If you’re sick and you could help others by enrolling in a clinical trial, why wouldn’t you want to help?”
Resources
- Learn more about the PREDICT trial
- Gastroenterology and Hepatology Program
- Helping Kids with IBD Take Control of Their Lives
- Services offered at South Clinic

