
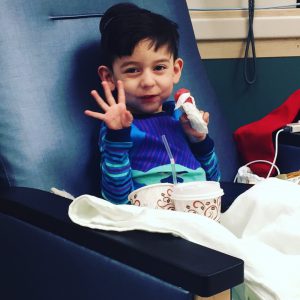
Spinal Muscular Atrophy (SMA) had progressively taken away the strength of 3-year-old twin brothers Harper and Hendrix to lift a cup of water, crawl or even take a deep breath on their own. Without access to a breakthrough treatment for the incurable genetic condition, the regression of their motor skills was certain to continue, potentially to the point that it was life-threatening.
So moments like the one that unfolded between Harper and Hendrix in a Seattle Children’s recovery room shortly after their fourth infusion of the new drug, Spinraza, represented much more than brotherly play to their parents, Crystal and Noe Ramos.
Harper raised his right arm high above his head and paused briefly before snapping it down in front of him as he released a makeshift ball of medical tape and paper. The object bounced and then skidded on the floor before it came to rest near Hendrix, who gave it a casual glance before returning his attention to the iPad in his lap he gripped firmly with his fingers.
Harper smiled.
A short throw. The tight grasp of an object. They are major accomplishments considered medically improbable prior to the treatment that has given the Ramos family newfound hope for the future.
“This treatment is helping give our kids a shot at a better childhood,” Crystal said. “They weren’t strong enough to even lift their arms above their shoulders two months ago. Now it’s incredible they can throw a ball.”
Signs of worry lead to grim diagnoses for Harper and Hendrix
The Ramos’ became increasingly concerned when the twins weren’t bearing any weight on their feet at 8 months old. Their oldest son, Santana, was walking when he was 10 months old.
“We started looking really closely at the different things they were doing and not doing,” Crystal said. “We knew something wasn’t right. We had a feeling of what it was but didn’t want to believe it.”
At 18 months old, Hendrix still was not crawling. Harper had lost his ability to crawl with just his arms. After numerous visits to doctors and physical and occupational therapy sessions, the family was referred to Seattle Children’s in March 2015.
On July 23, 2015, eight days before his second birthday, Harper was diagnosed with SMA Type II. Hendrix received the same diagnosis exactly one month later.
“We cried for a couple weeks but realized Harper and Hendrix needed us more than ever,” Crystal said. “We were going to give them whatever support they needed.”
A debilitating condition
SMA affects 1 in every 10,000 children in the United States and is the leading genetic cause of death among infants in the country. The condition impacts motor neurons, the nerve cells located in the spine that control muscles in the body. It can take away the ability to walk, move or breathe in severe cases. Children with SMA Type II are not able to walk and can have severely impaired motor function that requires lifelong assistance.
“SMA is a devastating diagnosis for parents and children,” said Dr. Susan Apkon, director of rehabilitation medicine at Seattle Children’s. “It is a condition known to continually progress, adversely impacting those affected and has life-threatening complications.”
Hope is found in a new treatment
The Ramos’ continued taking Harper and Hendrix to occupational and physical therapy sessions that did little to stem the tide of SMA. Eventually, the twins were weakened to the point they needed a cough assist machine to clear congestion in their lungs.
In looking for other answers, the family heard about trials featuring nusinersen, one of which was at Seattle Children’s, but the twins were unfortunately ineligible for the trial due to their age. It was disheartening as they heard from other SMA families that the drug was making unforeseen advances against the condition, like helping children walk.
However, the Ramos’ hopes lifted on Christmas Day 2016 , when they learned from their home in Pasco that nusinersen was approved two days earlier by the Food and Drug Administration and would be available commercially under the name Spinraza.
“It was the best present ever,” Crystal said. “We sat there laughing and crying. It was a really special moment.”
Since Seattle Children’s served as a clinical site for the trials, it had the operational knowledge and ability to become one of the first hospitals in the country to offer Spinraza. Soon after hearing the news, Crystal called Apkon, who provided a timeline for treatment .
The need for Medicaid
As a new drug for an orphan disease, the cost of Spinraza has garnered as much attention from insurance providers and media as has its unprecedented affect on SMA.
“While the potential benefit to patients is promising, it’s an expensive medicine. Immediately, physicians and families knew this was going to be a challenge,” said Apkon, who has treated and seen the devastating affects of SMA on patients for more than 20 years.
Each dose and spinal tap infusion of Spinraza costs more than double the median household income in Washington state. Six doses of the drug are required for the first year of treatment, costing about $750,000, followed by four doses each year thereafter. With twins, the cost was doubled for the Ramos’. Their private insurance covered a sizable portion of treatment but still left the single-income family of five in financial stress and agonizing over the reality that they may not be able to afford the treatment for the twins.
“Even when insurance agrees to cover the cost of this treatment for families, there is no guarantee of how much will actually be covered,” Apkon said.
Several other Pacific Northwest families seeking the new SMA treatment encountered similar financial hardship, even with private insurance coverage. Leadership at Seattle Children’s turned to Apple Health, Washington state’s Medicaid program, to inquire about additional financial support for families who needed the treatment. Nearly two-thirds of Washington’s Medicaid population is children.
Apkon was invited to discuss SMA and the benefits of the new treatment at the state capital in Olympia during Spinraza’s formal drug review process and Medicaid policy presentation. To the Ramos’ relief, Medicaid agreed to cover the costs of the medication, making it possible for Harper and Hendrix to benefit from the life-changing treatment.
“Without Medicaid support, it wouldn’t be possible for my children to benefit from this treatment that gives them the opportunity to have a childhood,” Crystal said. “The thought of ever not having Medicaid there to assist our family in the future is absolutely terrifying.”
Finding strength, reaching new milestones
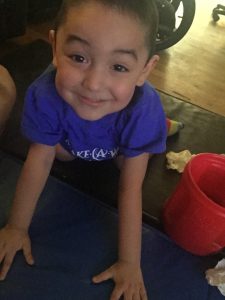
The progress Harper and Hendrix have made since they started Spinraza treatment at Seattle Children’s on February 22, 2017, has been remarkable. Both boys were able to sit in a chair without a harness three days after their second treatment. In March, Hendrix moved his leg forward in a walker for the first time since he was 8 months old. Harper crawled for the first time in more than two years in April, and Hendrix got into crawling position for the first time ever.
Their tremendous progress can be seen on the Harper & Hendrix Strong Facebook page. They laugh in pool workouts and spin in their wheel chairs. They splash in the water with their big brother. One video shows Hendrix unscrewing the cap of a sports drink before lifting the bottle to his lips.
But earlier posts on the page serve as stark reminders of what life was like before treatment and reasons why the Ramos family shares its story.
“There are so many children who are affected by SMA,” Crystal said. “I want to make sure as many children receive this treatment as possible. Everyone deserves the chance to be a kid.”
The American Health Care Act (AHCA) that was passed in the House of Representatives puts the health of the nation’s children, like Harper and Hendrix, at risk. The bill jeopardizes healthcare access for the more than 30 million children in the country who rely on Medicaid. Join Seattle Children’s in urging your elected officials to oppose healthcare reform that threatens children’s health.
Resources:
- Office of Financial Management – Washington state
- Cure SMA
- Harper & Hendrix Strong Facebook page
- Medicaid Facts – Washington – American Association of Pediatrics

