
Dr. David Rawlings and Dr. Andrew Scharenberg, researchers at the Center for Immunity and Immunotherapy at Seattle Children’s Research Institute, published a paper today in Science Translational Medicine that reveals a groundbreaking approach to engineering human T cells, which are crucial for fighting infection and show promise in treating autoimmune disorders, blood diseases and some types of cancer. Using HIV as a model for this new gene editing technique, they found that T cells could be engineered to resist HIV infection and simultaneously kill HIV-infected T cells or B cell tumors.
On the Pulse sat down with Dr. Rawlings to discuss this major discovery in gene editing and learn how it could help scientists working to find cures for HIV, cancer and other diseases.
Q: Tell us a little about your study and what you were trying to accomplish.
A: This study showcases a new and very efficient approach to gene editing. Genes are the basic building blocks of DNA. They tell a cell how to behave and what to do. Our research focuses on curing diseases by editing a patient’s genes to get them to behave a certain way, or to block a gene that’s causing a disease.
This study focused on gene editing in T cells, a critical component of the immune system. In HIV patients, those cells are disabled. We edited the T cell genome and gave it the ability to do two things simultaneously: Resist HIV infection and either kill HIV-infected cells or kill tumors.
Q: Can you tell us what a T cell is and why you use HIV as a model for this gene editing research?
A: T cells are white blood cells in the immune system that fight infection. HIV infects a person’s T cells and kills them, making that person susceptible to infections that a normal-functioning immune system would fight off. T cells have receptors on their surface, and the most common strain of HIV infects T cells by binding to a receptor known as CCR5.
We tested our new gene editing approach on the gene that controls CCR5 because there is a broad body of research around that receptor and scientists around the world are targeting it for HIV treatments, so our finding could have immediate applications.
Q: The T cells you engineered were able to kill HIV-infected T cells, resist HIV infection and kill tumors. How did you make them do that?
A: A T cell has 30,000 genes. We targeted the gene that controls the T cell receptor we focused on, named CCR5. We used a specific enzyme designed to make a cut in the CCR5 gene, which damaged the gene and eliminated the receptor for HIV. Simultaneously, to provide HIV- or tumor-killing capability, we placed a new gene with this function directly into the edited CCR5 gene, giving the edited T cells those new abilities. Our findings show that it is possible to edit the CCR5 gene so the T cell can both resist and kill HIV.
Another exciting discovery we made was the ability to place the edited genes exactly where we want them in the genome, which is a major advance. We make a cut in the DNA with a specific enzyme and use a non-infectious virus as a shuttle to place the new genetic material right where we want it. This allows us to precisely locate and control expression of the new gene.
Q: How efficient was this method? How often did the T cells work as planned after you completed the gene editing?
A: We used healthy T cells from more than 25 human donors and achieved about 60% efficiency in editing. That means 60% of the cells we edited were able to perform the actions we sought— HIV resistance plus HIV- or tumor-killing. Before this discovery, the best efficiency in editing any target gene in human T cells was in the single digits.
This is the first time anyone has made T cells do these things as efficiently as we did, so this is a major breakthrough.
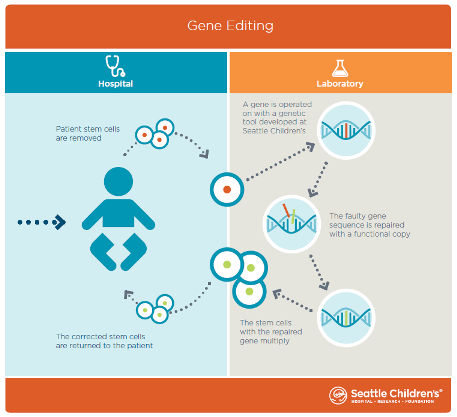
It’s also important to note that T cells reproduce. By having this high rate of efficiency, we will be able to take a patient’s T cells, edit them, expand them to large numbers and then send them back into the body where they would go to work for that person’s immune system. While not tested in our study, we expect these cells would continue to reproduce, potentially protecting that patient for a long period of time.
Q: Could this gene editing approach be used in other cells besides T cells?
A: Yes. In fact, that is one of our long-term goals. CCR5 is just one of many genes and HIV is one of many potential applications where this new technology will be useful. We have already used this approach successfully with more than five other genes.
Q: What’s the next step in this research?
A: In our study, we used this approach successfully in blood stem cells, which make T cells and all other types of blood cells. We are now optimizing the technique for that application. From an overarching perspective, this discovery will be useful for researchers working to cure cancer, autoimmune diseases and blood diseases such as severe combined immune deficiency and sickle cell disease.
Resources
- Study in Science Translational Medicine: Efficient modification of CCR5 in primary human hematopoietic cells using a megaTAL nuclease and AAV donor template
- Seattle Children’s Research Institute Faces of Research: Dr. Andrew Scharenberg
- Center for Immunity and Immunotherapies, Seattle Children’s Research Institute
- Seattle Children’s Research Institute Teams Up With bluebird bio to Pioneer Genome Editing and Gene Therapy Research in Pediatric Diseases

