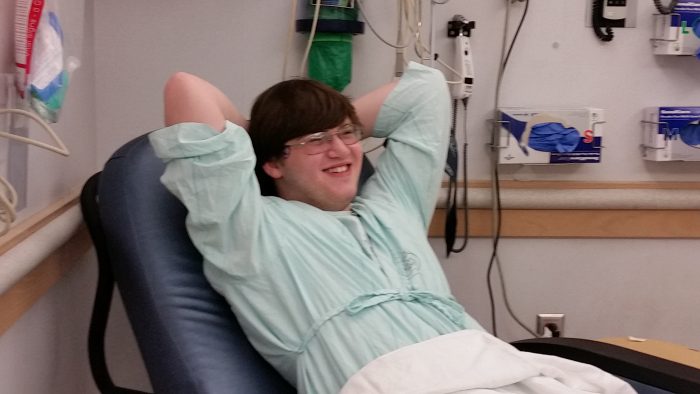
More than 50 research studies to understand, detect, treat and prevent the coronavirus in children and families have launched at Seattle Children’s since the virus emerged in late 2019. This is the first post in a new weekly series called “Quest(ion) for Discovery” highlighting this research in progress and the search for answers that could […]
In the largest genetic study of the most common birth defects of the brain diagnosed during pregnancy, researchers from Seattle Children’s Research Institute say their findings evolve our understanding of brain development. The findings will also change the information given to expecting parents when cerebellar malformations, such as Dandy-Walker malformation and cerebellar hypoplasia, are detected […]
Strokes come in many shapes and sizes. In children and adults, strokes often present sudden limb or facial numbness, confusion and dizziness. But some strokes that cause clots to develop in the small blood vessels of the brain don’t exhibit any symptoms at all. Studies have shown that hundreds to thousands of these small, asymptomatic […]
Wanting to do something different to address the alarmingly high number of newborn deaths in low income countries, Dr. Rachel Umoren, a neonatologist at Seattle Children’s, turned to virtual reality (VR). As mobile phone-based VR programs became increasingly accessible, Umoren thought the emerging technology could offer a better way to equip health care providers with […]
Every year, over 1.2 million people continue to be infected by HIV. Of these, about 160,000 are infants that contract the virus from their mother. Without treatment, a third of these infants die by their first birthday, and half of them do not make it to age 2. Thanks to programs that identify and treat […]
The first findings to result from a collaboration between Seattle Children’s Research Institute and Microsoft data scientists provides expecting mothers new information about how smoking before and during pregnancy contributes to the risk of an infant dying suddenly and unexpectedly before their first birthday. According to the study published in Pediatrics, any amount of smoking […]
For the first time in his life, Shanahan “Shanny” Dameral, 19, has a girlfriend. Soon, he’ll be graduating with a high school diploma and looking for his first job on the Kitsap Peninsula. What seems routine for many is a big deal for Shanahan and other children living with treatment-resistant or intractable epilepsy. For reasons […]
Recalling the treatments her daughter has had over the past year for an aggressive, very rare type of brain tumor known as atypical teratoid rhabdoid tumor, or ATRT, Audrey Taylor says it’s like watching a game where your favorite sports team keeps losing and then regaining the lead. “There are so many times when you […]
In a paper published in Nature Immunology, scientists from Seattle Children’s Research Institute reveal how a rare group of white blood cells called basophils play an important role in the immune response to a bacterial infection, preventing the development of sepsis. Researchers say their findings could lead to better ways to prevent the dangerous immune […]
With the countdown to the New Year almost here, On the Pulse caught up with a few of the patients who inspired our readers with their stories throughout 2018. Below, they offer their hopes, dreams and goals for the year to come. A shoulder above his cancer, Miguel sets sights on giving back In 2018, […]